IISc-incubated Shanmukha Innovations develops India’s first diagnostic kit for Sickle-Cell Anaemia
February 27, 2023 | Monday | News
Provides an affordable and efficient solution to screen large populations
image credit- shutterstock
Shanmukha Innovations, a health-tech startup incubated by the Society for Innovation and Development (SID) at the Indian Institute of Science (IISc) in Bengaluru, has developed SickleCert, India’s first indigenous diagnostic kit for Sickle-Cell Anaemia. A screening test kit called SickleFind has also been developed.
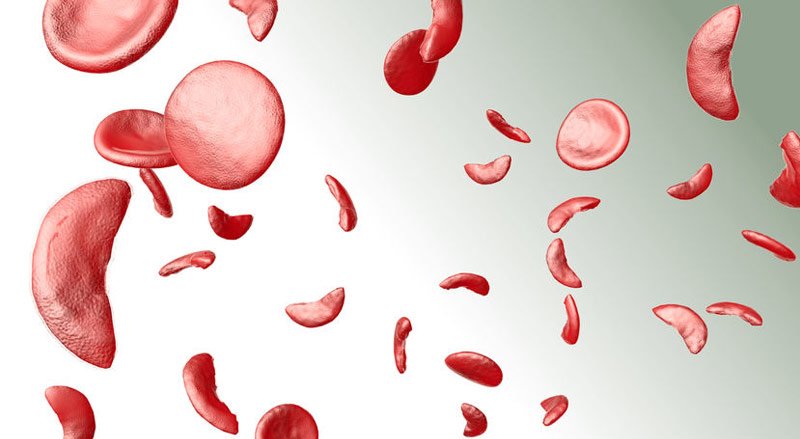
Sickle-Cell Anaemia is caused by a defective gene, Haemoglobin S, which causes flexible red blood cells to become stiff sickle-shaped cells, obstructing blood flow and increasing the risk of organ damage.
The technology called High Performance Optical Spectroscopy (HPOS) consisting of test kits (SickleCert), portable analyser (HaemoCube) was developed by researchers at IISc. The technology has been licensed to a spin-off from IISc, ShanMukha Innovations for commercialisation. A companion smartphone application enables digital data recording, analysis and integration with various registries, Ayushman Bharat Health Account (ABHA) and other cloud-based patient health record systems.
The Government of India has announced an ambitious National Sickle Cell Elimination Mission to eradicate the disease from the country by 2047 in the recent Budget. The Mission plans to screen 7 crore people over the next 3 years as a first step.
“With a tiny amount of blood from a finger prick, the test provides highly accurate results in 15 minutes. In batch mode over 40 samples can be tested in an hour enabling a large volume of screenings to be completed in a much shorter time span” says Prof. Sai Siva Gorthi, Principal Investigator, lead inventor of the technology and Associate Professor at the Instrumentation and Applied Physics Department, IISc.
The Central Drugs Standard Control Organisation (CDSCO) had approved the manufacturing license for SickleCert kits for ShanMukha Innovations as a Class C in-vitro diagnostic medical device in November 2022. In February this year, multi-centric field studies were successfully carried out as prescribed by the Indian Council of Medical Research (ICMR) across tribal districts in Madhya Pradesh and Maharashtra.
The tests yielded an accuracy measure of 97.6%, with a sensitivity of 96.9% and a specificity of 98.6%.