 Department of Pharmaceuticals launches Meditech Stackathon 2024 in collaboration with CII
Department of Pharmaceuticals launches Meditech Stackathon 2024 in collaboration with CII
 Maharashtra government gives re-recognition to medical courses at Colleges of Physicians and Surgeons, Mumbai
Maharashtra government gives re-recognition to medical courses at Colleges of Physicians and Surgeons, Mumbai
 Health Ministry unveils National Action Plan for prevention and control of snakebite envenoming
Health Ministry unveils National Action Plan for prevention and control of snakebite envenoming
 Artemis Medicare Services raises Rs 330 Cr to bolster healthcare footprint
Artemis Medicare Services raises Rs 330 Cr to bolster healthcare footprint
 Kotak Strategic Situations India Fund II invests Rs 400 Cr in Biorad Medisys
Kotak Strategic Situations India Fund II invests Rs 400 Cr in Biorad Medisys
 Indegene gets oversubscription by 7.35 times on day 2
Indegene gets oversubscription by 7.35 times on day 2
 ABLE inks MoU with Emerging Biopharmaceutical Manufacturers Network
ABLE inks MoU with Emerging Biopharmaceutical Manufacturers Network
 Serum Institute of India partners with ImmunityBio for global supply of BCG across all cancer types
Serum Institute of India partners with ImmunityBio for global supply of BCG across all cancer types
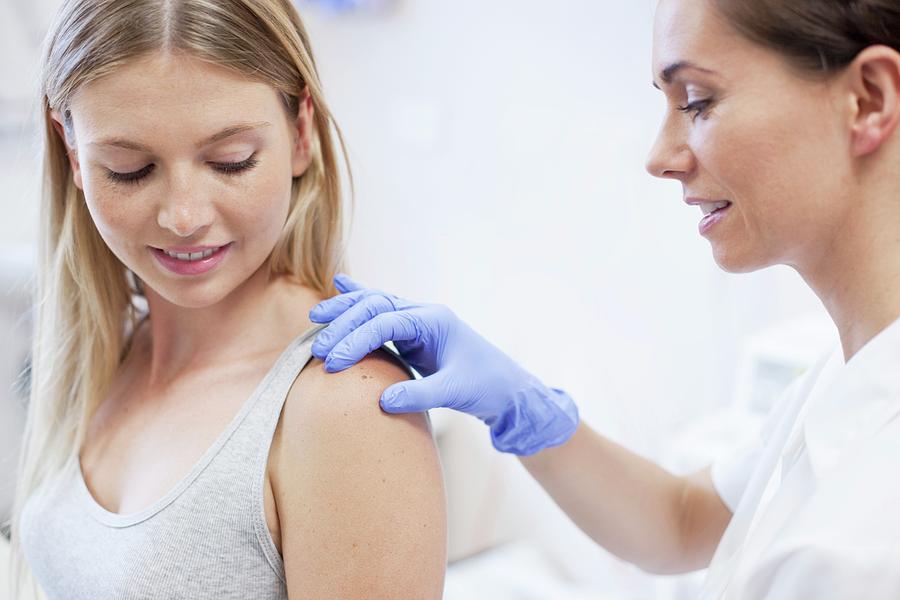 India Ramps Up HPV Vax Efforts
India Ramps Up HPV Vax Efforts
 WHO prequalifies Takeda's dengue vaccine
WHO prequalifies Takeda's dengue vaccine
 EPA, FDA, and USDA issue joint regulatory plan for biotechnology
EPA, FDA, and USDA issue joint regulatory plan for biotechnology
 Major step in malaria prevention as three West African countries roll out vaccine
Major step in malaria prevention as three West African countries roll out vaccine

Science | 15 May 2024 | News
Infosys Science Foundation announces new direction for Infosys Prize
The Infosys Science Foundation (ISF) has announced that it is steering the Infosys Prize in a new di...Read more

Opinion | 01 May 2024 | Views
How do you evaluate the growth scenario of the Indian market specifically for laboratory and analytical instru...Read more

MedTech | 09 May 2024 | News
Omron Healthcare collaborates with AliveCor to improve heart healthcare in India
Omron Healthcare India, the Indian arm of the Japanese global leader in medical equipment for home health moni...Read more

Digital Healthcare | 16 May 2024 | News
Doceree unlocks untapped potential of digital platforms in healthcare
Delhi-based startup Doceree has announced the launch of its highly anticipated High Interaction & Engageme...Read more

Supplier | 16 May 2024 | News
Sartorius to advance drug discovery and manufacturing with AI in collaboration with NVIDIA
Germany-headquartered life science group Sartorius is expanding its multidisciplinary collaboration ...Read more

People | 16 May 2024 | News
Takeda appoints Annapurna Das as General Manager for India Operations
Takeda Biopharmaceuticals India (formerly known as Baxalta Bioscience India Private Limited), part of a global...Read more

Academics | 13 May 2024 | News
Dr Kiran Mazumdar-Shaw inaugurates bio sciences lab at Chanakya University, Bengaluru
Chanakya University, in Bengaluru, recently celebrated a momentous stride in scientific innovation with the in...Read more

Text blogs | 05 January 2024 | Views
Suffering From GIGO? Turn to GRDs Instead, for better healthcare innovations
Here’s the Problem… Let me get straight to the point. Many pharma and life sciences companies th...Read more

India’s Life Science Instrumentation Industry:A Disruptor with Global Ambitions?
The Indian laboratory and analytical instrument industry is on the brink of a transformative journey, propelled by rapid technological advancements and a growing emphasis on indigenous innovation. With the convergence of cutting-edge technologies such as Artificial Intelligence, Machine Learning, Big Data Analytics, Online and Atline testing, Cloud computing, and Process Analytical Technologies, the industry is poised for accelerated growth and expansion. Experts from different fields of the industry have highlighted the burgeoning opportunities for domestic players in the laboratory equipment and analytical instrument segment, exploring the evolving landscape, challenges, and strategies for fostering innovation and competitiveness.
For Feedback, please email us at: communications@mmactiv.com